AAVnerGene Announces AAVone®2.1, a Next-Generation Single-Plasmid AAV Production Platform Achieving ~1e16 GC/L and >70% Full Capsids at Harvest
PR Newswire
ROCKVILLE, Md., May 8, 2026
ROCKVILLE, Md., May 8, 2026 /PRNewswire/ — USA — AAVnerGene Inc. today announced the launch of AAVone®2.1, the next-generation evolution of its proprietary AAVone® single-plasmid AAV production system, designed to improve AAV vector productivity, full-capsid yield, manufacturing efficiency, and cost effectiveness.
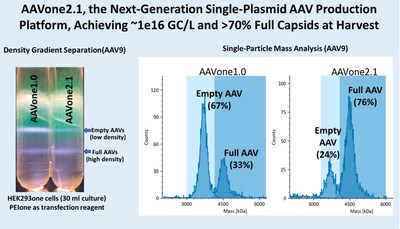
AAVone®2.1 has achieved ~1×1016 genome copies (GC)/L of cell culture and greater than 70% full capsids at harvest, representing a significant advance over AAVone®1.0 and conventional multi-plasmid AAV production systems, which often generate high proportions of empty capsids. The platform has demonstrated performance across multiple AAV serotypes and compatibility with HEK293-based cell lines and existing manufacturing workflows.
These improvements may reduce plasmid requirements, culture volume, purification burden, processing steps, processing time, and manufacturing cost, while improving overall vector quality. By addressing key constraints in AAV production, AAVone®2.1 is designed to support the clinical and commercial scalability of AAV-based gene therapy programs.
“AAV manufacturing remains one of the most important technical and economic challenges in gene therapy,” said Qizhao Wang, Ph.D., Chief Technology Officer of AAVnerGene. “Conventional multi-plasmid systems have enabled the field for decades, but limitations in productivity, empty capsid burden, and purification complexity continue to create barriers to development and commercialization. AAVone®2.1 was developed to address these challenges through a simplified single-plasmid system that improves vector productivity and full-capsid ratio. “
“AAV gene therapy continues to face commercialization challenges, with manufacturing cost, vector quality, and process scalability among the most critical constraints,” said Daozhan Yu, PhD, Chief Executive Officer of AAVnerGene. “AAVone®2.1 has the potential to make AAV manufacturing simpler, more scalable, and more cost-efficient, helping more AAV gene therapy programs become clinically and commercially viable.”
AAVnerGene’s partners have licensed the technology and are developing multiple AAV gene therapy programs using the platform. The company’s goal is to provide enabling technologies that help overcome manufacturing barriers, reduce development costs, and expand the number of gene therapy programs that can reach patients.
About AAVnerGene Inc.
AAVnerGene is a Maryland-based biotech company developing solutions to address bottlenecks in AAV gene therapy, including manufacturing, toxicity, qualification, and delivery. Its platforms include AAVone®, AAV-Q (a potency and rcAAV assay platform), and ATHENA (a capsid engineering platform), designed to improve productivity, quality, scalability, efficiency, and safety in AAV gene therapy drug development.
Media & Business Inquiries:
customer@aavnergene.com
www.aavnergene.com
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/aavnergene-announces-aavone2-1–a-next-generation-single-plasmid-aav-production-platform-achieving-1e16-gcl-and-70-full-capsids-at-harvest-302766885.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/aavnergene-announces-aavone2-1–a-next-generation-single-plasmid-aav-production-platform-achieving-1e16-gcl-and-70-full-capsids-at-harvest-302766885.html
SOURCE AAVnerGene Inc.